Japan Approves First Self-Amplifying mRNA for COVID in Adults
I wrote about samRNA a few months ago and now it is happening
At the end of November, Japan's Ministry of Health, Labour and Welfare approved CSL and Arcturus Therapeutics' ARCT-154, the first self-amplifying mRNA (samRNA) vaccine for COVID for initial vaccination and booster for adults 18 years and older.
Source: https://www.nature.com/articles/d41586-023-03859-w
The approval was based on positive clinical data from several ARCT-154 studies, including an ongoing 16,000 subject efficacy study performed in Vietnam as well as a Phase 3 COVID-19 booster trial, which achieved higher immunogenicity results and a favorable safety profile compared to a standard mRNA COVID-19 vaccine comparator.
You read that correctly, the trial is ongoing meaning that it has not been completed and yet this injection has received approval. All study participants are being monitored until one year after their second injection so again, long-term effects are unknown. Not only that but the trial was an integrated phase 1/2/3a/3b, randomized, observer-blind trial in Vietnamese adults. This means they combined all phases of the clinical trials. As way of review, proper clinical trials for vaccine development can take 10 to 12 years. And the shortest period of time for vaccine approval was 4 years for the mumps vaccine in the 1960s. But maybe Japan has a different approval process than the US FDA approval process.
I wrote about this a few months ago and you can read about what samRNA is in more detail here:
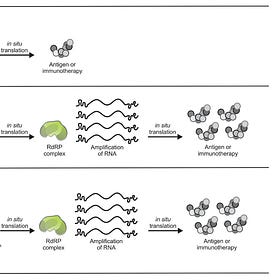
Just as a reminder samRNA jabs go a step further than conventional mRNA by integrating the genes needed for the replication and synthesis of the antigen-encoding RNA, effectively establishing a biological printing press for fabricating the vaccine inside cells. Dr. Mobeen Syed does a great job in this video explaining how samRNA works inside a host cell.
People often think that the samRNA vaccine platform is simply a variation on conventional mRNA shots, “but in practice it’s really not”, says Anna Blakney, a bioengineer who studies the technology at the University of British Columbia in Vancouver, Canada. “samRNA is a totally different beast.”
They admit samRNA “vaccine” platform does have some downsides. samRNA also engages with the immune system in intricate ways — for example, by forming replication intermediates that help to stimulate beneficial immune-signaling pathways. However, excessive stimulation can backfire, including when the “vaccine” prompts the immune system to block RNA replication, thereby nullifying its benefits.
Safety, immunogenicity and efficacy of the samRNA ARCT-154 COVID gene therapy
Researchers reported the primary efficacy objective was met as two doses of ARCT-154 with an efficacy of 56.6% (95% CI: 48.7–63.3) against COVID-19 disease of any severity. They claim efficacy against severe COVID-19 was 100% in healthy 18–59-year-olds but this age group does not suffer severe COVID anyway, especially those considered as “healthy” meaning no underlying medical conditions.
In addition the side effect profile of the samRNA was the same as the conventional mRNA injections. This should be no surprise given the toxicity profile of the spike protein. I’m not certain how samRNA technology can be beneficial in any way against SARS-CoV-2 given all the data that has emerged about the harmful effects of spike protein on the cardiovascular and neurological systems not to mention on fertility and general inflammation in the body.
Nathaniel Wang, chief executive and co-founder of Replicate Bioscience in San Diego, California, a company that develops samRNA “vaccines” anticipates that, with continuing advancements, samRNA technology will increasingly replace conventional mRNA in a diverse array of therapeutic contexts.




When will they actually make a vaccine for sarscov2. Still waiting... in the meantime let’s all keep cutting corners by measuring severity of infection and try new technologies that make things more confusing. Meanwhile wastewater monitoring shows infections are as high as ever. If spike via infection we know is bad for us, how would it be any different for self-replicating encoded spike. And there is still the nasty issue of nanogram quantities of loose plasmid vector needed to make these things. For $2.1M you can attempt to correct sickle cell disease...CRISPR also requires a gRNA to direct to the editing site...how many times has this been tested in clinical trials? So many companies getting millions in investment for LNP RNA therapies. Genetic editing tech floodgates are open.