Regulatory Capture Continues to Plague FDA Even After Reforms Were Suggested in "The Truth About the Drug Companies" in 2004
Three years of pandemic corruption has brought a windfall for failing pharma companies who have enjoyed the largest government bailout at the public’s expense with no end in sight.
The term “regulatory capture” has been thrown around a lot in the last three years. In 2004 Dr. Marcia Angell, former editor-in-chief of the New England Journal of Medicine (NEJM), published an exposè on corruption in the pharmaceutical industry and its financial ties with the regulatory agency, the FDA. The relationship between big pharma and the FDA personifies regulatory capture which has gone unabated in the nearly twenty years since Angell recommended reforms in The Truth About the Drug Companies: How They Deceive Us and What To Do About It.
Dr. Marica Angell is a nationally recognized authority in the field of health policy and medical ethics.
Dr. Marcia Angell studied microbiology as a Fulbright Scholar before receiving her medical degree from Boston University School of Medicine in 1967. She is a board certified pathologist and a member of such prestigious organizations as the Institute of Medicine, the National Academy of Sciences and is a fellow of the American College of Physicians.
In 1997, Time magazine named Dr. Marcia Angell one of the 25 most influential Americans for that year. After 20 years with the NEJM, Dr. Angell became the first woman editor-in-chief of this premier journal of medical science in 1999. She then went on to become a senior lecturer at Harvard Medical School’s Department of Global Health and Social Medicine. Dr. Angell has devoted her life to researching, writing and speaking on topics incorporating medical ethics, health policy, the nature of medical evidence, the interface of medicine and the law, and end-of-life care.
Angell was perfectly positioned to speak about corruption in the pharmaceutical industry.
From her post at NEJM Angell had a front-row seat on the appalling spectacle of the pharmaceutical industry and became a fierce critic of the pharmaceutical industry. She saw them gain nearly limitless influence over medical research, education, and how doctors do their jobs. This culminated in publication of her book titled The Truth About Drug Companies in 2004.
Dr. Angell witnessed clinical trials sponsored by the companies that made the drugs which ensured that they had control over how the trials were designed, how the data was analyzed and where/when it would be published.
What was most troubling was the change in the relationship between academic medicine and clinical research for the drug companies which introduced more bias into the research. There was a change in the terms and conditions with the research itself as pharma companies became wealthier. Researchers at academic and medical institutions that were paid to perform trials were given less control over the work sometimes disallowing researchers to see the data they had collected. One of the worst forms of bias was that pharma companies would not permit researchers to publish negative results and would bury that data. However, all clinical trials MUST by law be submitted to FDA for approval and it MUST INCLUDE ALL THE DATA from all the trials. As if this isn’t bad enough, the more shocking thing about this is that in some cases FDA would help bury negative data and move the drug along for approval anyway.
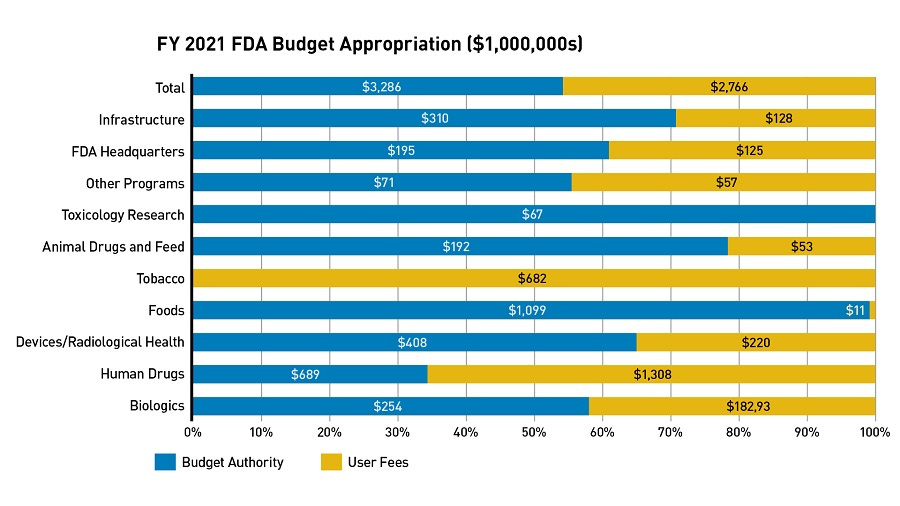
Another troubling change she witnessed came when the 1992 Prescription Drug User Fee Act was passed allowing the FDA to collect fees from drug manufacturers to fund the new drug approval process mainly to expedite the review of drug and supplement applications.
Angell stated, “It's time to take the Food and Drug Administration back from the drug companies...In effect, the user fee act put the FDA on the payroll of the industry it regulates. Last year, the fees came to about $300 million, which the companies recoup many times over by getting their drugs to market faster.” (February 26, 2007 Taking back the FDA)
In 2021 industry user fees covered around 65 percent of the human drug approval process. An estimated $796,065,980 in prescription drug user fees were reportedly collected for the period October 1, 2014, through September 30, 2015 by the FDA. This amount ballooned to well over 1.3 billion dollars by 2021 for human drugs. In fact, as part of the approval process for its COVID-19 vaccine, Pfizer made a wire transfer to the FDA of $2,875,842 million in May 2021 with EUA approval of its injection following shortly afterwards in August 2021.
The biggest issue with these user fees is that the public are the end users and not the FDA.
Source: https://www.fda.gov/about-fda/fda-basics/fact-sheet-fda-glance
Angell proposed reforms to redirect pharma companies back to their original mission.
Dr. Angell proposed a program of vital reforms, which included restoring impartiality to clinical research and severing the ties between drug companies and medical education.
Her proposal addressed seven broad problems that she discussed in her book (page 239).
Drug companies produce too many me-too drugs and too few innovated ones.
FDA is too much in the grips of the industry it regulates.
Drug companies have too much control over clinical research of their own products.
Patents and other exclusive marketing rights are undesirably long and too elastic.
Drug companies have too much influence over medical education about their own products.
Important information R&D, marketing, and pricing is kept secret.
Prices are too high and too variable.
Angell proposed changes that would help to shorten exclusive marketing rights which would affect profits but more importantly the ability of the industry to influence government and the FDA. Her suggested changes would work to bring better drugs at lower prices and loosen the grip of pharma on public policy and the medical profession.
Pharma companies attacked Angell on her claims.
As Angell’s stinging criticisms gained resonance, the pharma industry hit back. In 2012, John LaMattina published a hit piece in Forbes titled “Marcia Angell's Attacks on Pharma Have Lost all Credibility”.
LaMattina claimed, “The problem with Angell’s arguments is that they are rife with inaccuracies and fallacies. Furthermore, she makes no accounting for changes in the industry that have occurred over the last decade.”
LaMattina had a massive conflict of interest; he was the president of Pfizer Global Research and Development in 2007, a fact that should have disqualified him from writing on the subject. He certainly had bias when it comes to this topic. He offered little to no evidence to back up his allegation that the claims she made were false. For instance, in response to her claim that pharma does not produce innovate drugs, he stated that 2012 had proven to be a great year for patients with the approval of 35 innovative drugs. However, not all these drugs came from pharma as biotech companies had major drugs approved. This aligns with what Angell outlined in her book that the real source of most innovative drugs are universities and biotech companies.
LaMattina said that the attack he found to be “particularly offensive is that on health standards”.
“Angell implies that medical experts are being paid off by pharmaceutical companies to influence national health guidelines to favor the greater use of drugs. This is a very dangerous accusation, because it suggests that unnecessary drugs are being prescribed.”
Now that we are working our way through the opioid crisis we can see that this has been proven correct. People were being prescribed opioids for everything from a hangnail to back pain and most disturbing of all they were told by pharma marketing agents that the drugs were not addictive. Roughly 500,000 people in the U.S. have died from opioid overdoses since the opioid crisis began in 1999. Not surprisingly, Pfizer was among the list of top 10 largest federal settlements list for the opioid settlements.
And now we have all witnessed the largest government bail out of pharma companies with the COVID-19 pandemic and roll out of genetic vaccines.
Since the pandemic was declared in 2020, medical doctors were disincentivized to prescribe inexpensive, safe, and widely available drugs for the treatment of COVID-19. Not only were drugs pulled from the shelf, pharmacists were told not to fill prescriptions for certain drugs and clinicians were threatened with loss of their job and/or license if they used said drugs to treat patients. There is the belief that this was done to allow for the roll out of new mRNA genetic vaccines.
One pharma company that benefitted in a big way from these actions was Moderna, whose name came from a combination of “modified” and “RNA”. Prior to the pandemic this company was floundering as it had not brought a single product to market since its creation in 2010. Investors barely had interest in the company, which had yet to produce a medicine. Things changed in 2020 with the initiation of Operation Warp Speed, the public–private partnership initiated by the United States government to facilitate and accelerate the development, manufacturing, and distribution of COVID-19 vaccines, therapeutics, and diagnostics.
Moncef Saloui, then a member of the board of directors of Moderna, stepped up to head Operation Warp Speed. It’s no wonder Moderna genetic vaccine was selected to move forward as the second candidate vaccine to receive EUA approval by the FDA. Once Moderna’s mRNA genetic vaccine received EUA approval Saloui stepped down as head of Operation Warp Speed. Not only did Moderna receive an infusion of capital, receiving $483 million from Operation Warp Speed, but the company went on to make billions in profits from its genetic vaccine. In essence, Saloui helped to line his own pockets with millions in profits gained by pushing his company’s injection to the head of the line for approval.
Source: https://www.fiercepharma.com/pharma/moderna-covid-vax-scarfed-sales-184b-2022-company-says
Given all this, I think it is safe to say that reforms are still needed in the biopharmaceutical medical industry complex. Dr. Marcia Angell has previously pointed out ‘the real battle in healthcare is one of truth versus money’. It is well past time to uncouple truth from money. We need to get the FDA off the industry’s payroll and afforded proper public support to carry out its mission of protecting the public from big business, not the other way around.







100% they are captured. Yes, yes, and yes.